
Verzenio + an aromatase inhibitor
Explore the effectiveness and dosing of Verzenio + an aromatase inhibitor.
How effective is Verzenio + an aromatase inhibitor (AI)?
Verzenio was studied in combination with an aromatase inhibitor as initial endocrine-based therapy in 493 postmenopausal women with newly diagnosed hormone receptor positive (HR+), human epidermal growth factor receptor 2 negative (HER2–) advanced or metastatic breast cancer (MBC). 328 patients were treated with Verzenio + aromatase inhibitor and 165 were treated with an aromatase inhibitor alone.
Clinical trials are still ongoing to determine if there is an overall survival benefit with Verzenio.

Proven to help women live significantly longer without disease progression
Verzenio + an AI delayed disease progression for a median of 28.2 months vs 14.8 months with an AI alone.

More than half of the women saw their tumors shrink
55.4% of women on Verzenio + an AI saw their tumors shrink vs 40.2% on an AI alone.

Complete response: no detectable signs of cancer
3.4% of women on Verzenio + an AI had a complete response, or no detectable sign of cancer, vs 0% with an AI alone.

Partial response: tumor shrinkage of 30% or more
52.1% of women on Verzenio + an AI had a partial response with tumor shrinkage of 30% or more vs 40.2% with an AI alone.
SELECT SAFETY INFORMATION
Verzenio may cause serious side effects, including:
Diarrhea is common with Verzenio, may be severe and may cause dehydration or infection. The most common time to develop diarrhea is during the first month of Verzenio treatment. If you develop diarrhea during treatment, your healthcare provider may tell you to temporarily stop taking it, stop your treatment, or decrease your dose. If you have any loose stools, start taking an antidiarrheal medicine (such as loperamide), drink more fluids, and tell your healthcare provider right away.
Taking Verzenio + AI
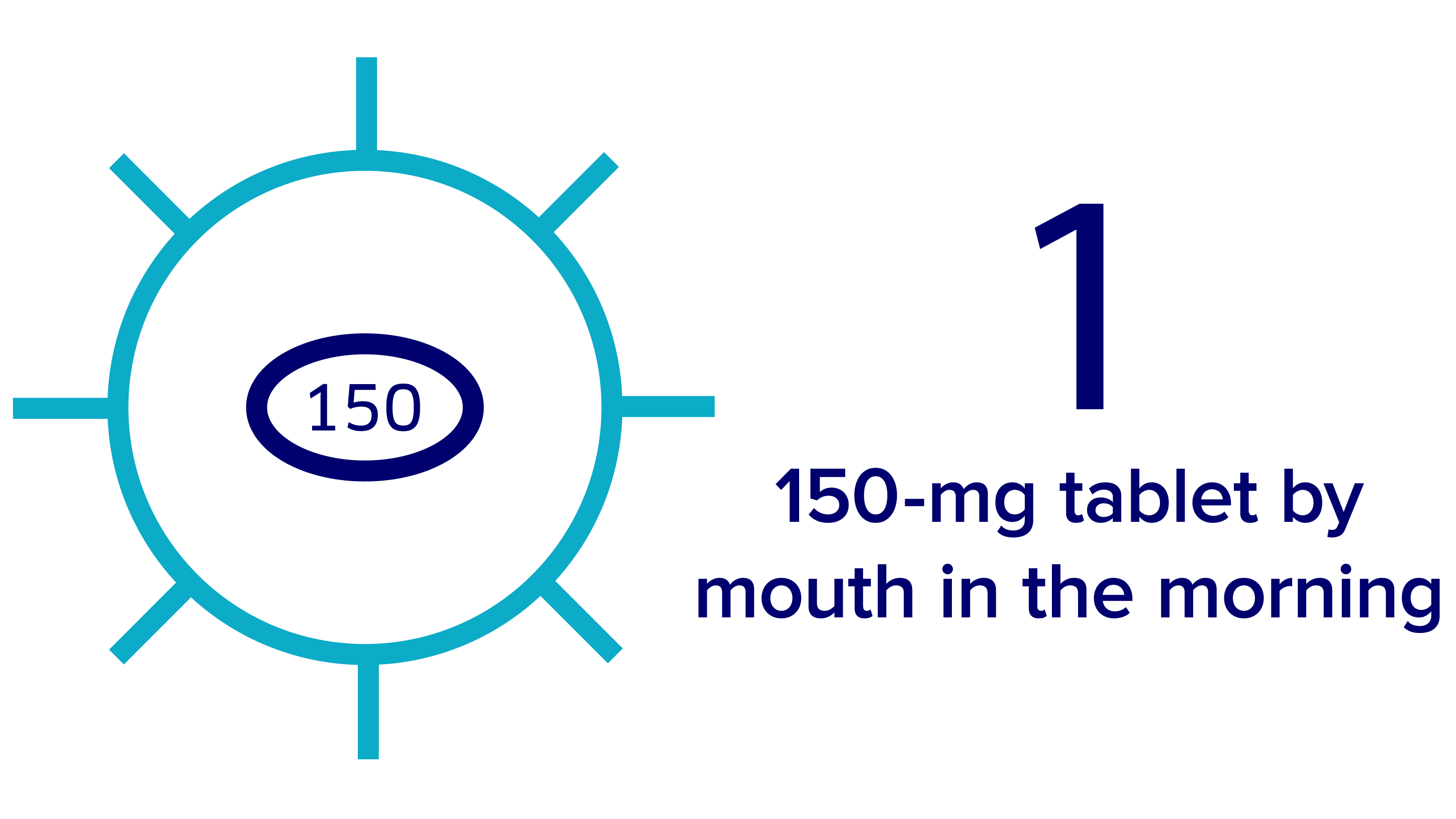
The recommended dose of Verzenio in combination with an AI is 150 mg by mouth twice a day, taken once in the morning and once at night.

You will receive 4 individual packs, each one containing your dose of 150-mg tablets for 7 days. Each of these packs includes 14 individual 150-mg tablets.
- Multiple tablet strengths (50 mg, 100 mg, and 150 mg) allow your doctor to make dose adjustments based on your tolerance
- If you need to reduce your dose due to tolerability, Verzenio is approved to be dosed as low as 50 mg twice daily, as directed by your doctor
- No matter what dose you are prescribed, you will take 1 tablet, 2 times a day, unless your doctor tells you otherwise
If your doctor prescribes:

50-mg dose
You will receive
4 red packs.

100-mg dose
You will receive
4 purple packs.

150-mg dose
You will receive
4 light blue packs.

More time without cancer getting worse
Verzenio + an AI delayed disease progression for a median of 28.2 months vs 14.8 months with an AI alone.
Addressing side effectsIf you have questions about Verzenio, or need more information on how to take it, you should talk to your doctor. You can also call us at 1-844-VERZENIO (1-844-837-9364) or sign up for our Verzenio Continuous Care™ Support Program.
Learn about Savings & Support, including how to enroll in the Verzenio Continuous Care Program